1. The AMIDE EUTECTICs (0.33K+0.67Na)NH 2
as new inorganic proton conductor
at 310 – 400 K in solid state
The conductivity 10 mS/cm at 363 K aws measured in closed double-chamber electrochemical cell
(+)C | (0.33K+0.67Na)NH 2 | Sn(-)
Pt witres were used as referencr electrodes/
The remarkable sel-diffusion of hydrogen atoms aws observed by protium-deuterium exchange between eutectics under study and gaseous mixture ammonia and molecular hydrogen.
2. Self-supporting behavior of unusual circuit of three batteries based on hydroxide proton conductors: including opportunity to work as solar cell or hydrogen fuel cell.
↓↑{C | KOH.2H2O |Sn===C | KOH.2H2O |Ge} ↓↑
↓↑ ↓↑
↓↑++++++++++{C | KOH.2H2O |TiFe} +++++↓↑
On the end of this cell (indicated here by ARROWS) at room temperatures the voltage near 1400 mV remain more than one year (!) and returned after producind the current up yo o.1 mA
3. HALL EFFECT of PROTON conductivity of SOLID HYDRATE of POTASSIUM HYDROXIDE
Tags:
February 22nd, 2013
admin
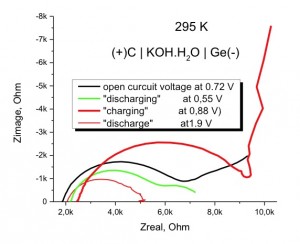
(-) Ge | KOH⋅H2O | C(+)
Solid protonic conductors based on crystallohydrates of alkaline metals are studied from 2007. Our attention is concentrated on KOH⋅nH2O (0,5≤ n ≤2). Meltng points are 100°C (n=0.5), 146°C (n=1), 42°C(n=2.0). These particular points of phase diagram KOH – H2O are interesting first of all for basic research, but intermediate ‘n’ have to be studied from applied viewpoint owimg to composite formation at low temperatures and high proton conductivity )more 1 mS/cm. However to use these compounds on elect traditional electrode material –graphite – and rochemical devices of any type it is necessary to find electrode materials compatible with solid hydroxide compounds. On this site metallic Pd, Ti, TiFe and Sn are already presented together with traditional electrode material –graphite. As Carbon and Tin are the memebers of IV group of periodical table we are starting to study the classical semiconductors Si an Ge.
The electrochemical cell under study was formed in Teflon tube of 10 mm in diameter. Electrodes were plate p-Ge (28 Ohm.cm) and C(graphite rod). . The interelectrode distance was 1 cm.
The electrolyte was KOH⋅1.05 H2O (125°C<T crystallzation <147°C). Such mixture has been simplified the procedure of the formation of cell.
The resistance of electrolyte was 1.3 kOhm. According to impedance spectroscopy the resistance of whole cell was 5-7 kOhm at low current density (<0,1 mA/cm2).
The Open Curcuit Voltage of this cell was 0,72 V at 300 K and varied with tempature as 1 mV/K.
Cermanium was the negative electrode.
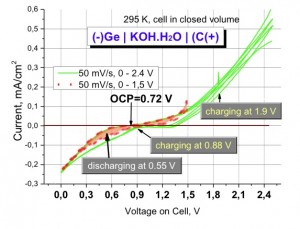
Using Germanium ans Carbon allows to consider the opportunity:
- to combine with Solar Cells,
- to use as low-drain power sources in microelectronics.
Tags:
November 12th, 2012
admin
CsHSO4 (cesium hydrosulfate) was the first acidic salt possessing high-temperature modification of very high protonic conductivity (A.I.Baranov et al 1982). Now this acidic salt y as a member of the huge family of proton-conducting acidic salts, is considered as a touchstone to study the opportunity to use these family as an electrolyte in electrochemical devices. In this case an electrolyte has to have not only high conductivity, but also to be compatible with different electrode materials. Earlier we (Yu.M.Baikov et al 2006 etc) have revealed that solid.hydroxide protonic conducrtors (e.g. KOH.nH2O, n=1, 2) possess the high enough electrochemical activity even at room temperatures in heterostructures with graphite and tin (See this site.) It is important that they are not noble metals like Pt, Pd !!.
Recently we have synthesized novel heterostructure (-)Sn | CsHSO4 | C(+) and revealed that it is really electrochemical cell having stable e.m.f. near 0.7 V at temperature higher 450 K i.d. after phase transition in CsHSO4 . Such cells can work as a battery. Electrodes from tin plate and graphite rod without special treatment (!) provide the current density more than 1 mA|cm2.
From fundamental viewpoint it is very important to compare of the eletrochemical behavior of two distinquished anions: basic OH- (proton acceptor) and acidic HSO4- (proton donor).
Tags:

ISE 63 Prague
Let’s consider new promising family of electrochemical devices
-
Batteries
-
Rechargeable batteries
-
Fuel Cells
Based on new perspective solid hydroxide protonic conducters.

SSPC-16 Grenoble
Tags:
INORGANIC PROTONIC CONDUCTOR
at LOW TEMPERATURE UP TO – 500C (220 K) (!!!)
ELECTROCHREMICALLY ACTIVE WITHOUT USING NOBLE METALLS
PROTONIC HETEROJUNCTION “TiFeX|KOH·2H2O”
we’ve been presented in 2012 on
i) , EMRS 2012 May 2012 Strasbourg France,
ii) Fast Ionic Transport-10 Chernogolovka Moscow district 1-4uly 2012
iii) Fundamental Problems of Solid Ionics-11 Chernogolovka Moscow district 5-8 July 2012
iv) ISE_63 Annual meeting 19-24 August 2012 Prague Czech Republik
v) Solid State Proton Conductors SSPC_16 10-14 September 2012 Grenoble France
Tags:
Dear Colleagues!
Please consider and discuss below presented сomplete quasichemical description of some cubic oxides similar YO1.5 and double perovskites in water and oxygen atmosphere
Read more…
Tags:
I would like to propose for starters to divide the set of subjects onto two groups according to remarks in “Introduction” about preferably my personal interest (items ”a” and “b”) and evidently common interest (items “c” and “d”). Of course such division has enough relative character, but, by my opinion, it seems suitable.
Read more…
Tags:
Yu. M. Baikov, E. I. Nikulin, B. T. Melekh, L. G. Baikova
The family of complex materials such as crystalline hydrates and solid eutectics, on the base of hydroxides of alkaline metals (Na, K, Rb, Cs) has been studied thoroughly from the general physico-chemical point of view. However, the high proton conductivity of the members of this family has been revealed on for the last few years. Correspondingly there was no information on the electrochemical activity of heterostructures “alkaline proton | electron conductor”. In our opinion the crucial reason for this objectionable approach of world ionic community to whole alkaline-hydroxide-family was due to dramatic history of arising and then falling of interest in individual alkaline hydroxides. The key aim of the paper is to present our results of the development of solid and molten hydroxide materials which are good proton conductors at intermediate / room temperatures (450 < T < 250K) and low-humidity or even anhydrous conditions, as well as are electrochemically active in MEA with Ti, TiFe, Sn and Si. The main experimental results and informal discussions on necessary further progress in fundamental understanding of the underlying proton conduction mechanisms are collected now on website http://www.solidionic.com.
Authors : Yu. M. Baikov, E. I. Nikulin, B. T. Melekh, V. A. Klimov
“Old” isotopic methods, namely both isotopic exchange (IsEx) and isotopic effect(IsEff), are the effective characterization technique at the study of interface and surface processes at different length scales. Factually, they are in-situ techniques of significant importance at the study of mass and charge transport across and along interfaces of recently discovered ionic conductors on the base of alkaline hydroxides, namely crystalline KOH·H2O (Tmelt=146°C) and KOH·2H2O (Tmelt=42°C) as individual compounds and solid eutectic NaOH+KOH (ENaK) (Tmelt=185°C). See http://www.solidionic.com. The isotopic technique supplied to traditional physico- and electrochemical techniques is factually phenomenological modelling for a deeper understanding of the underlying transport and reactivity mechanisms. We present the results of the study of IsEff (H<=>D) of both the conductivity and EMF of the heterojunction electrode|protonics as well as IsEx “gas-electrolyte” and “electrode-electrolyte”. The rate of IsEx “gas(H2)-a.m.electrolytes” was compared with the exchange-current on the interface “TiHx(or PdHy)| a.m.electrolytes”. The main conclusions are following. i)The mobility of hydrogen species is higher along internal interfaces than across them in the microheterogenic ENaK. ii) The reality of hydrogen heterojunction has been confirmed. iii) The surface of hydroxides itself can activate the molecular hydrogen.
Comparison of electrochemical cells with different isotoppic conten in electrolyte
C(grahite)|KOH·2H2O|Sn

Isotopic effect of voltammetry
Tags:




